Poisonous Mushroom Compound Could Help Flow Batteries
By Katherine Bourzac, for C&EN

As utility companies adopt more renewable sources of energy, the need for large-scale energy storage on electrical grids is growing. Solar, wind, and other renewables produce electricity intermittently, so storage is key to making those sources practical. One way to meet this need is with flow batteries, which store energy in large tanks of chemicals. But for this technology to compete with conventional lithium-ion batteries, the storage capacity of flow batteries must be increased, and their stability improved.
In a conventional battery, lithium and other ions move back and forth between solid electrodes. However, redox flow batteries store energy in the oxidation state of a charge carrier, commonly vanadium, which stays put. And rather than an ion moving back in forth, protons and electrons or hydroxide ions migrate across a membrane that separates tanks of solvents and the charge carrier.
A group of chemists working to improve the flow battery has found inspiration in an unusual place: the forest floor. At the Spring 2019 American Chemical Society national meeting in Orlando, Patrick Cappillino, a chemist at the University of Massachusetts Dartmouth, described how his group has adapted a compound made by mushrooms to improve the stability of flow batteries.
Vanadium is the charge carrier of choice because it can be chemically toggled between several oxidation states, Cappillino said. To prevent the ions from moving across the membrane, V4+ is held in place by a chemical carrier. A good vanadium-binding compound has to do many things. It should have high affinity for the metal ion. And it should be very stable—these batteries need to be charged thousands of times and last for years. These compounds also need to be highly soluble in solvents that can withstand high voltages without decomposing. The higher the voltage, the more energy the flow battery can store. Existing flow batteries are limited to about 1 V, and that voltage needs to increase four- or five-fold for the technology to be a contender, Cappillino said. (Lithium ion batteries operate at about 3.8 V.)
Cappillino was surprised to learn from a colleague that mushrooms in the Amanita genus, which includes several poisonous fungi such as toadstools, accumulate relatively large amounts of vanadium in a coordination complex called amavadin. Amavadin has the highest vanadium-binding stability ever measured. Cappillino wondered if he could adapt it to work in a flow battery.
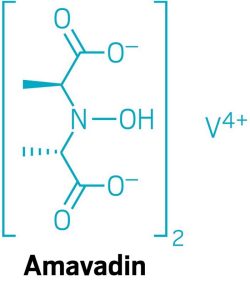
The University of Massachusetts, Dartmouth, researchers reported a 100-gram synthesis of an amavadin-inspired compound called VBH2- two years ago (J. Mater. Chem. A 2017, DOI: 10.1039/C7TA00365J). Yang-Shao Horn, a chemist who develops vanadium redox flow batteries at the Massachusetts Institute of Technology, is impressed by the synthesis method and the molecular design of the compound. At the national meeting, Cappillino discussed his group’s progress on translating the fungi-inspired compound to flow batteries. VBH2- is highly selective for vanadium, and the compound is stable. But it’s not soluble enough in high-voltage solvents for use in a flow battery. The Dartmouth researchers are now working on tuning the material’s solubility.