Don’t Sweat It: How Moisture-Wicking Fabrics Keep You Cool and Dry
If you visit your local sporting goods store, you’ll be met with a dizzying array of choices: performance apparel, technical clothing, moisture-wicking fabrics. Sixty dollars for a shirt to sweat in? What gives?
Welcome to the world of fabric technology. In 2020, people worldwide spent 160 billion dollars on sportswear. The goal is not just to look good but to feel good too. Compression garments massage and support muscles, improving blood flow. Running shoes offer abundant arch support. Sweat-wicking fabrics are designed to keep you cool and dry. Do they really work? Are they worth the cost?
If you go for a run on a hot day in a cotton shirt, it will quickly become soaked. While cotton fabrics may be soft and comfortable, cotton is pretty hydrophilic. (Place a few drops of water on a cotton ball and see what happens—the cotton soaks it up like a sponge.) Looking like you jumped into a swimming pool with your clothes on is not a good look. Plus, a sweat-saturated shirt can cause no small amount of irritation, chafing, and even bleeding in sensitive areas.
Textile engineers have coined a term for how much water a fabric retains when wet: the moisture regain value. This value is found by dividing the mass of water in a saturated fabric by the mass of the dry fabric. If a 100 g sample of fabric has a mass of 110 g when wet, for example, its moisture regain would be 10% (mass of water/mass of dry fabric).
Here are moisture regain values for some common fabrics:
Cotton ≫ 8.5% | Wool ≫ 16% | Cotton-polyester (50/50) ≫ 4.45% | Nylon ≫ 4% | Polyester ≫ 0.4%
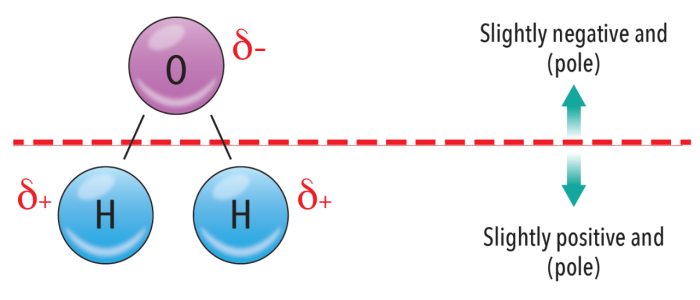
The ability of fabrics to absorb water is largely a result of their polarity. Water is polar, with electrons being drawn toward the more electronegative oxygen.
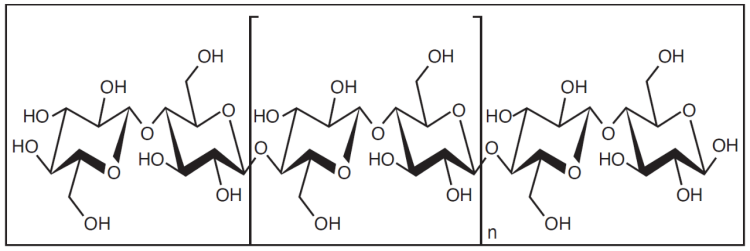
Cotton is primarily made of cellulose, a polymer composed of glucose units (C6H10O5) strongly linked together. Although strongly hydrophilic, cellulose is not water soluble; otherwise that cotton shirt would dissolve when it got wet. However, the -OH’s (hydroxyl groups) from the cellulose stick out of the backbone, like burrs on a dog’s tail. It is these highly polar hydroxyl groups that have such a strong affinity for water.
In nonpolar substances, the London dispersion forces that act between nonpolar molecules tend to be weaker than the dipole-dipole forces that act between polar molecules. Thus, nonpolar compounds are more hydrophobic. Oil, being nonpolar, does not dissolve in water. Dry cleaners are adept at removing stains—most of which are nonpolar (think grease stains)—because they have access to a wide array of nonpolar solvents.
If we don’t want to work out in a hydrophilic fabric, do we want to work out in a hydrophobic one? Well, if you’ve ever weathered a storm in an old-fashioned rubber raincoat, you soon realized you were just as wet on the inside as on the outside. Since your perspiration has nowhere to go, it just lays against the skin, making you feel clammy and gross. If you place a drop of water on a fabric and it is repelled, demonstrating its hydrophobicity, it is most definitely not sweat wicking.
Sweat-wicking fabrics work as a result of capillary action—the tendency of a liquid to move upward in a narrow tube or space, in apparent defiance of gravity. Capillary action works because water molecules both adhere and cohere to one another.
Cohesion involves the attraction of like things to one another. Each polar molecule essentially acts like a tiny magnet, with the negative side of one molecule joining up with the positive side of another. Water droplets form when billions and billions of water molecules—each one highly polar—clump together.
Adhesion, in which unlike things stick together, is a tad more complicated. Water droplets that stick to a windowpane are demonstrating adhesion. Since glass is electrically neutral, it may seem odd that water would stick to it. Everyone knows opposite charges attract, but why would something be attracted to a neutral?
Adhesion works because of the action of dipole-induced dipole intermolecular forces of attraction. As the positive side of a polar molecule approaches a nonpolar molecule, the electron cloud of the nonpolar species shifts slightly toward the polar molecule. Since a dipole has been induced in the nonpolar molecule, it is instantly attracted to the polar molecule.
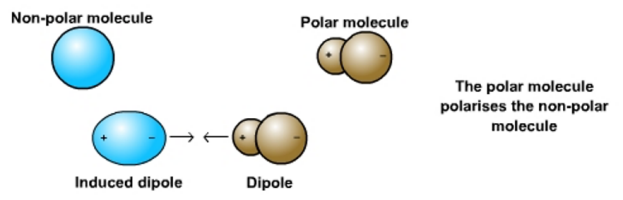
Capillary action occurs when the forces of adhesion are stronger than the forces of cohesion. Because water molecules have a stronger attraction to the sides of a narrow tube than to one another, they creep upward.
The first step in designing a sweat-wicking fabric is to find a fiber that will facilitate capillary action. A fiber is the base material from which clothing is made, and it can be natural or synthetic. If animal hair is being used, a single hair acts as a fiber. If a bunch of fibers are twisted together, you have a strand of yarn. When strands of yarn are woven together, you have fabric. Fabric is then cut and sewn together to make the clothes we wear.
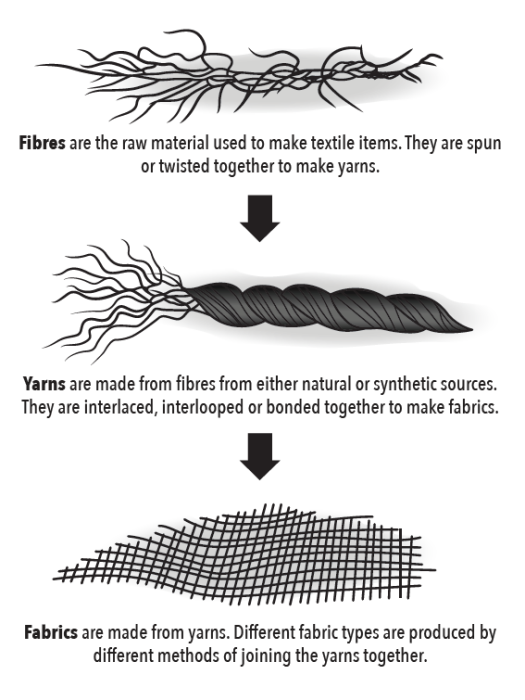
It is important that the fiber used not be too hydrophilic, like cotton, which won’t wick away moisture at all. But purely hydrophobic fibers that repel water won’t work either. In order for capillary action to occur, there must be some attraction to water—just not too much.
One popular fabric used in sweat-wicking clothing is polyester. Being petroleum based, polyester is quite hydrophobic, with a moisture regain of only 0.4%. To make it a little less so, it can be chemically treated with a hydrophilic coating. Alternatively, the polyester fibers can be interwoven with hydrophilic fibers, forming a blend, creating an excellent moisture-wicking garment.
Nylon is another popular choice for sweat-wicking clothing. Nylon is polyamide, and the amide units comprising it are polar. Nitrogen is less electronegative than oxygen, so amide bonds in nylon aren't as polar as the hydroxides in cotton. As a consequence nylon is not as hydrophilic as cotton, but it is hydrophilic enough to wick away sweat.
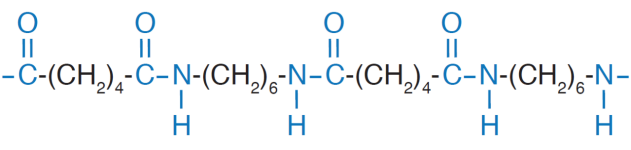
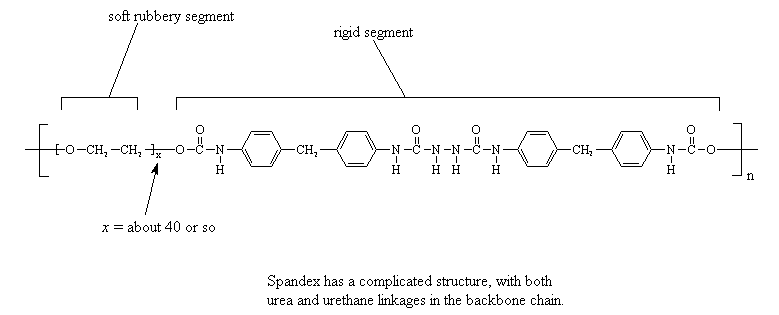
Spandex, the stretchy fabric commonly found in leggings and bike shorts, only has moderate wicking ability. You will never find a garment made of pure spandex, however—it is usually used in conjunction with nylon or polyester. So not only do your yoga pants allow for freedom of movement but they also keep you dry, even during intense workouts.
Some types of sweat-wicking clothing have an inner layer next to the skin that is hydrophobic and an outer layer that is hydrophilic, creating a push/pull effect. The hydrophobic layer repels water, and so the moisture is pushed upward into the hydrophilic layer, where it can evaporate.
Wool, especially merino wool, has excellent moisture-wicking properties. Wool fibers are hydrophilic on the inside but hydrophobic on the outside due to the presence of lanolin, a waxy material secreted by the sheep’s sebaceous glands, which makes their coats repel water.
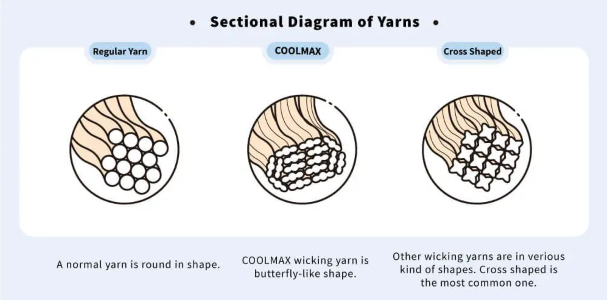
Once the appropriate material is chosen, the next step is to weave the yarn together in such a way as to create the sweat-wicking effect. This step requires the most ingenuity. Most yarns have a circular cross section. When woven together, they fit snugly. Since there is little room for water to travel between the strands, their ability to wick moisture is limited.
But if a fabric can be constructed in such a way as to create spaces between the strands, then moisture can travel through these spaces via capillary action. This amazing little feat of engineering is accomplished by constructing yarns with noncircular cross sections—they can be triangular, cross-shaped, or some other oddball shape.
When these strands of yarn are placed together, they do not fit snugly. There is space between them. These little spaces create micropores, which are just the right size to facilitate capillary action. Moisture can freely travel through these pores to the outer surface, where almost like magic, it is whisked away.
The magic doesn’t happen, though, without a lot of rigorous testing. Before ever hitting the market, a fabric will undergo a battery of tests to make sure it will perform as predicted. These experiments are easy enough to perform yourself—all you need is water and some samples of fabric.
In a vertical wicking test, two strips of cloth are suspended with their ends in water. The fabric in which water travels the fastest will have the greatest wicking ability.
In a horizontal wicking test, a predetermined amount of water is dripped onto the fabric and its horizontal spread is measured. The fabric that spreads the most in the shortest time wins.
A transverse wicking test most closely mimics what a moisture-wicking garment is designed to do. Water is absorbed from below the fabric, and its rate of spread through the fabric and then along its surface is measured. The faster the spread, the better.
For clothing to keep you cool and dry, once sweat reaches the surface of the garment it must evaporate. Sweating itself does not cool you—it’s the evaporation of sweat that cools you. Since evaporation is endothermic, it must absorb energy. When liquid water vaporizes, hydrogen bonds are broken, and breaking bonds always requires an input of energy. This energy is drawn from your body, cooling you down.
Your clothing should help you stay cool, but not too cool. If your clothing hasn’t effectively wicked away moisture but has rather left it saturated with sweat, you can cool down too much once your workout is over as this sweat begins to evaporate. Because you are no longer moving you may feel chilled, especially in cooler weather.
The select interplay between chemistry and engineering has revolutionized clothing. You no longer need to feel hot during your workout and cold afterward. With sweat-wicking gear, you can feel just right all the time.
References
A Multi Directional Wicking Instrument to Measure Wicking Characteristics of Fabrics Under Dynamic Movements
https://www.researchgate.net/publication/327712897_Wicking_Instrument
[Accessed May 2022].
Engineering of High-Performance Textiles
https://books.google.com.HighPerformanceTextiles
[Accessed May 2022].
Liquid transport across fabric layers
https://www.researchgate.net/publication/240721054_Liquid_Transport_in_Fabric_Structures
[Accessed May 2022].
This article is adapted from ChemMatters Magazine. The orginal story was published on October 1, 2022.